Molecules in 2D containers: in control of polymorphism
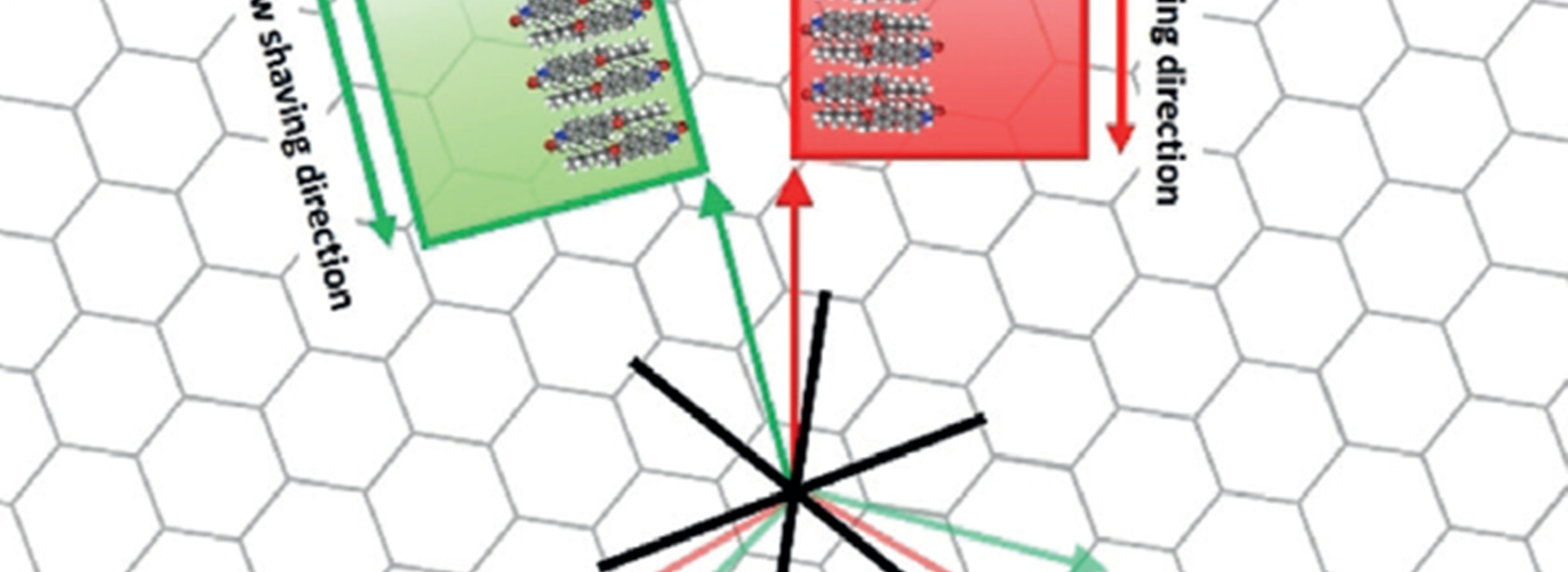
Control over polymorph formation in the crystallization of organic molecules remains a huge scientific challenge. Now, preferential formation is presented of one polymorph, formed by chiral molecules, in controlled twodimensional (2D) nanoconfinement conditions at a liquid–solid interface. So-called nanocorrals to control concomitant polymorph formation were created in situ via a nanoshaving protocol at the interface between 1-phenyloctane and covalently modified highly-oriented pyrolytic graphite (HOPG). The preferentially formed polymorphs, which were less stable in the large-scale monolayers, could be selected simply by varying the orientation of the square nanocorrals with respect to the HOPG lattice.
Read more about this in this paper.
To enable comments sign up for a Disqus account and enter your Disqus shortname in the Articulate node settings.


